Cite this
Phase Diagrams (and Pseudosections) for Petrologists
Standard phase diagrams are graphical representations of the equilibrium relationships between minerals (or others phases). These relationships are governed by the laws of thermodynamics. Standard phase diagrams show how phases or phase assemblages change as a function of temperature, pressure, phase composition, or combinations of these variables.
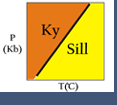
Some phase diagrams (those for 1-component diagrams) depict relationships involving multiple phases having the same composition (for example, the relationships between the vapor, liquid and solid forms of H2O). Other diagrams (such as the one shown on the right), depict the relationships between a number of compounds having different compositions. Still others show how compositions of phases change under different conditions.
Pseudosections are a type of phase diagram that shows the fields of stability of different equilibrium mineral assemblages for a single bulk-rock composition. Standard phase diagrams may include many reactions but, depending on its composition, a particular rock may only experience a few (or none) of them. Pseudosections only include those reactions that affect a particular composition. Fields on a pseudosection are labeled (specifying the equilibrium mineral assemblage) with the reaction lines unlabeled (although the specific reaction can be deduced).
Phase Diagram Archive
This is a diverse collection of phase diagrams, intended to be used by instructors when teaching petrology or mineralogy classes.
Besides single page diagrams, we also provide animations for some of the more useful systems. The animations are multi-page pdf files.
Binary Igneous Diagrams
These temperature-composition (TX) diagrams show melting relationships, and some subsolidus relationships, involving two chemical components. Some involve incongruent melting; most do not. Some of these diagrams involve eutectic and peritectic relationships.
Download binary phase diagrams.
Ternary Igneous Diagrams
Ternary igneous phase diagrams are triangular diagrams that show melting relationships involving three chemical components. The diagrams may involve eutectic, peritectic and cotectic relationships.
Download ternary phase diagrams.
Metamorphic P-T Phase Diagrams
Natural metamorphic systems may involve many chemical components. To simplify analysis and interpretation, petrologists may study model systems containing only a few components. Some of the diagrams available here involve only a few components and a few phases. Others are more complex.
Phase diagrams that show many reactions that take place in a single chemical system are termed "petrogentic grids." Petrognetic grids generally involve more components and more reactions that simple model metamorphic systems. The idea is to include enough components and phases so that the grid may be used to interpret natural metamorphic mineral assemblages.
Download metamorphic P-T phase diagrams.
Metamorphic T-X Phase Diagrams
For some rock compositions, metamorphic assemblages vary greatly depending on the composition of the fluid present. Metamorphic fluids are generally dominated by H2O and CO2, and the ratio H2O:CO2 can control mineral stability. So, phase equilibria are plotted on T-X diagrams instead of P-T diagrams. (X refers to the mole fraction of 2 or H2O in the metamorphic fluid that is present.)
Download metamorphic T-X phase diagrams.
Other Phase Diagrams
The above examples are of phase diagrams commonly used by geologists. Phase diagrams are also useful in other fields, including chemistry and engineering.
- H2O (solid-liquid-vapor relationships)
- NaCl-KCl (eutectic and solvus involving alkali halides)
- CH3CH2OH-H2O ("ethanol")-H2O (diagram similar to plagioclase and olivine melting)
- C12H22O11 ("sucrose")-H2O (binary eutectic)
Web-based Resources
- Phase Rule and Binary Systems (more info) - John Winter, Whitman College, PowerPoint (Chapter 6)
- Two Component Phase Diagrams ( This site may be offline. ) - Steve Nelson, Tulane University
- Binary Phase Equilibria Tutorial ( This site may be offline. ) - Paul Asimow, CalTech
- Binary and Greater Systems (more info) - John Winter, Whitman College PowerPoint(Chapter 7)
- Ternary Phase Diagrams ( This site may be offline. ) - Steve Nelson, Tulane University
- Magma Diversification (more info) - John Winter PowerPoint (Chapter 11 - Magmatic Processes)
- Index of Animated Phase Diagrams (more info) - Ken Windom, Iowa State University
- Metamorphism of Calcareous and Ultramafic Rocks (more info) - John Winter PowerPoint (Chapter 29)
Problem Sets/Labs/Activities on Simple Phase Diagrams
- Problem Set: Phase Diagrams - Dexter Perkins, University of North Dakota
- An Introduction to T-X Phase Diagrams with problems - Dexter Perkins, University of North Dakota
- Useful Phase Diagrams - John Brady, Smith College
- Phase Diagrams from Kitchen Chemistry - John Brady, Smith College
- Binary Eutectic In-class Exercise (Di-An) - Alan Glazner, University of North Carolina at Chapel Hill
- Homework on Volcanic Rock Chemistry and Binary Phase Diagrams - Jane Selverstone, University of New Mexico
- Plagioclase Phase Diagram - Dexter Perkins, University of North Dakota
- Constructing a Two Component Phase Diagram Using Experimental Data in the Hypothetical System A-B - R. K. Smith, University of Texas at San Antonio
- Ternary System: Determination of Crystal-liquid and Crystal-crystal Proportions Using the Lever and Tangent Rules - R. K. Smith, University of Texas at San Antonio
- The Use of Visualization and Sketches of Thin Sections to Encourage a Better Understanding of Phase Diagrams: Binary and Ternary Phase Diagram Exercises - Jennifer M. Wenner, University of Wisconsin at Oshkosh and Drew S. Coleman, University of North Carolina
- An Isograd and Mixed-Volatile Exercise Using Data from the Ubehebe Peak Contact Aureole - William H. Peck, Colgate University and Gregory T. Roselle, Bechtel SAIC Company
- Metamorphism of Siliceous Dolostone: An Example from the Alta Stock, Utah - Cameron Davidson, Carleton College
- Interpreting T-X Diagrams - Dexter Perkins, University of North Dakota
- Lab Activity: Timing of Mineralization in the Palm of your Hand: Cross-cutting Relations, Copper Minerals and Ore-forming Hydrothermal Fluid Evolution (Activity-activity Diagrams) - Barb Dutrow, Louisiana State University