Advanced Modeling Programs: Theriak-Domino
What is it?
The Theriak-Domino routines are best known for their ability to calculate and plot equilibrium phase diagrams (also known as pseudosections). Theriak, the heart of the collection, calculates the stable mineral assemblage and equilibrium phase compositions for a given rock (bulk composition) at specified P, T conditions. Domino is the key program for plotting diagrams. Additional programs calculate thermodynamic functions, (pseudo)binary or (pseudo)ternary phase diagrams, equilibrium phase compositional isopleths, and density, volume or modal equilibrium distributions. The programs, including source code (Fortran 77), are available for both Mac and Windows. Konstantin Petrakakis (Department of Geological Sciences, University of Vienna, Austria) maintains the Windows version.
Hints to get going
The software can be dowloaded for free (link below) and can be installed and running in well under an hour. The many files and folders can be confusing, especially for students unfamiliar with computer programming. To install efficiently and keep things as simple as possible, you may wish to follow the installation and set up suggestions given here (Acrobat (PDF) 113kB Jun21 11). (Alternatively, just follow the instructions that you will get when you downlaod the software.) I also have include several worked examples with explanations of input and output.
Principles
Since the early 20th century, and seminal works by Goldschmidt and others, petrologists have known that the stability of minerals, and of mineral assemblages, may be evaluated by applying the principles of thermodynamics to natural systems. Goldschmidt demonstrated that, given the necessary thermodynamic data, it is possible to calculate phase diagrams showing where assemblage-limiting reactions occur in PT, TX, or PX space. For a long time, unfortunately, the requisite data were unavailable, and so experimentalists studied model systems and determined phase diagrams through painstaking experimentation.
Beginning about forty years ago, thermodynamic data sets, computer programs, and computer capabilities improved so that many calculations became routine. In 1975, Slaughter et al. produce an APL computer program that was widely used to calculate phase diagrams. Other programs soon followed, and capabilities improved. Today, petrologists use many different programs (e.g., TWQ) to calculate phase diagrams, and robust and internally consistent thermodynamic data sets are available for minerals, melts, and aqueous solutions.
Mineral and mineral-assemblage stability, however, depend on rock composition as well as on thermodynamics. So, beginning in the 1990's, petrologists revised or rewrote computer programs to take bulk composition into effect. For a given rock composition, at specified PT conditions, the programs calculate the equilibrium compositions of coexisting minerals and the relative amounts of each that will be present. The programs produce diagrams, sometimes termed pseudosections or equilibrium phase diagrams that look like phase diagrams but that are different. Phase diagrams show all reactions in a model system that could take place; equilibrium phase diagrams show stable mineral assemblages. The difference is that some reactions that appear on phase diagrams will not affect a rock of the wrong composition. For example, a reaction that involves forsterite will not affect a rock that contains no forsterite in the first place.
The Theriak-Domino package of de Capitani and coworkers is one of the more popular programs for calculating equilibrium phase diagrams, primarily because of ease of use. Other routines that serve the same purpose include: THERMOCALC (Powell et al. 1998; Powell and Holland 1994), Perplex (Connolly 1990), and Gibbs (Spear et al. 2001).
Applications
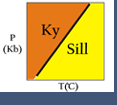
The Theriak-Domino routines can be used for a variety of purposes. Figure 1 (above) shows a classic equilibrium phase diagram (pseudosection) generated by Domino. The diagram shows stability fields for different mineral assemblages, for a specific model compositional system.
Figure 2 (left) shows a more complicated example, generated by Domino, involving a natural pelite composition and more than 20 phases. Fields are unlabeled, due to crowding, but Domino generates a complete list of reactions (Acrobat (PDF) 9kB Jun15 11) for reference.
Other, less conventional kinds of calculations that can be made using Theriak-Domino are shown in Figures 3 and 4 (from Perkins and Anthony, 2011; the data to make these figures came from Theriak but drafting was done using another program):
Strengths, limitations, and worked examples
Theriak-Domino has lots of things going for it. It is easy to obtain and install. Once installed, it is quite easy to figure out how to do a number of different kinds of calculations. (The users' guide is quite good.) It is more intuitive and easier to use than some other similar computer packages. The primary drawbacks are the same as for other, similar programs. (1) The accuracy of results depends on the accuracy of the database. And, there are still some questions about the thermodynamic properties of some minerals and, especially, of some mineral solutions. (2) Complicated calculations involving many components and phases may take a long time (>30 minutes) to complete.
For some information about setting up the programs, and worked examples to help clarify some of the input and output, click here (Acrobat (PDF) 113kB Jun21 11).
Problem Sets
- Problem Set: Theriak-Domino Exercise - Dexter Perkins, University of North Dakota; this is a problem set that has students calculate a standard PT diagram using TWQ and then compare it to a pseudosection generated by Theriak-Domino. It also involves using Excel.
- Problem Set: Making Good Looking Diagrams from Theriak-Domino Output - Dexter Perkins, University of North Dakota; this exercise, which is appropriate after students have completed the previous one listed, teaches students how to clean up computer-generated output to make it ready for reports or publications.
Supporting Resources: Powerpoint Presentations by Christian de Capitani, Theriak-Domino Short Course, Geoscience Canada Meeting, Calgary AB, May 2010
Start here! This is amost excellent set of presentations that provide clear and concise instructions with worked examples.
- Introduction to Theriak-Domino--Equilibrium Assemblages and Phase Diagrams (PowerPoint 7.8MB Jul24 11)
- Theriak Algorithm (PowerPoint 7.7MB Jul24 11)
- Domino Algorithm (PowerPoint 1.2MB Jul24 11)
- Domino--Getting Started (PowerPoint 4MB Jul24 11)
- T-D Flow Charts (PowerPoint 1.3MB Jul24 11)
- Theriak-Domino Input File--THERIN (PowerPoint 411kB Jul24 11)
- Example: Equilibrium assemblage diagrams: sample TN205 (PowerPoint 2.4MB Jul24 11)
- Scripts in Domino (PowerPoint 2.2MB Jul24 11)
- Defining a P-T-loop (PowerPoint 1.6MB Jul24 11)
- Activity H2O (PowerPoint 671kB Jul24 11)
- Buffers (PowerPoint 1.2MB Jul24 11)
- T-X CO2 (PowerPoint 3MB Jul24 11)
- Equilibrium assemblage calculations: a comparison (PowerPoint 2.4MB Jul24 11)
- AFM (PowerPoint 2.2MB Jul24 11)
References
de Capitani C. and Brown T.H. (1987): The computation of chemical equilibrium in complex systems containing non-ideal solutions. Geochim. Cosmochim. Acta 51:2639-2652.
de Capitani C. and Petrakakis K. (2010): The computation of equilibrium assemblage diagrams with Theriak/Domino software. American Mineralogist 95:1006-1016
de Capitani C. (1994): Gleichgewichts-Phasendiagramme: Theorie und Software. Beihefte zum European Journal of Mineralogy, 72. Jahrestagung der Deutschen Mineralogischen Gesellschaft, 6,48.
Perkins, D. and Anthony, E.Y. (2011) The evolution of spinel lherzolite xenoliths and the nature of the mantle at Kilbourne Hole, New Mexico. Contributions to Mineralogy and Petrology, in press.
Slaughter, J., Kerrick, D.M., Wall, V.J. (1976): APL computer programs for thermodynamic calculations of equilibria in P-T-XCO2 space. Contributions to Mineralogy and Petrology 54:157-171.